|
Some devices will have internal regulators or components because they run at a lower voltage, and you may be able to bypass them. These will have safety measures in them and should easily give a few hours at what you need.Īlternatively hack your device. You can find these offthe shelf with a usb Power delivery enabled power bank with a USB PD or Quick charge trigger module to get 9V out. 3LR12 (4.5-volt), D, C, AA, AAA, AAAA (1.5-volt), A23 (12-volt), PP3 (9-volt), CR2032 (3-volt), and LR44 (1.5-volt) batteries This is a list of the sizes, shapes, and general characteristics of some common primary and secondary battery types in household, automotive and light industrial use. Aka a nice lithium battery pack with 9V output. Ideally what you need is modern technology. That means you will get 7 minutes of battery life. Specialty Battery, Model: 76A/LR44 Battery Voltage: 1.5 V Battery Chemistry: Alkaline Battery Capacity: 130 mAh Min. From you can see that a 1 Amp draw for a good battery may only give 90 mAh of capacity and the voltage drops below 7V real quick. In reality the battery chemistry will reduce the voltage and current capacity as the current draw increases. Even if you could draw 1.5 Amps at 9V in an ideal setup, with a typical 500 mAh 9V alkaline battery, you would get less than a 3rd of an hour life on it. Different batteries have different discharge rates.9 volt batteries are designed for low current long life applications. Rechargeable batteries in the AA size are available in multiple chemistries: nickelcadmium (NiCd) with a capacity of roughly 6001,000 mAh, nickelmetal. Typically, the longer the discharge time, the more energy produced. When the battery is connected to a circuit, a chemical reaction takes place within the cell and current flows. This calculator-type battery is made of quartz and is ideal for small electronics like pH meters, calculators and. The alkaline chemistry behind these batteries makes them cheap and easily. It is the current supplied by the battery, measured in amperes, multiplied by the number of hours the battery can supply that amount of current. The electrodes are submersed in the electrolyte. The common disposable AA batteries have a starting voltage of 1.5 V. It’s actually quite complex The voltage produced by a battery depends on it’s chemical make-up. The discharge rate of batteries is expressed in ampere-hours. In very simple terms, they’re not always multiples of 1.5 or 1.2 volts. Higher voltages may be obtained by connecting several cells in series so that their voltages add together. 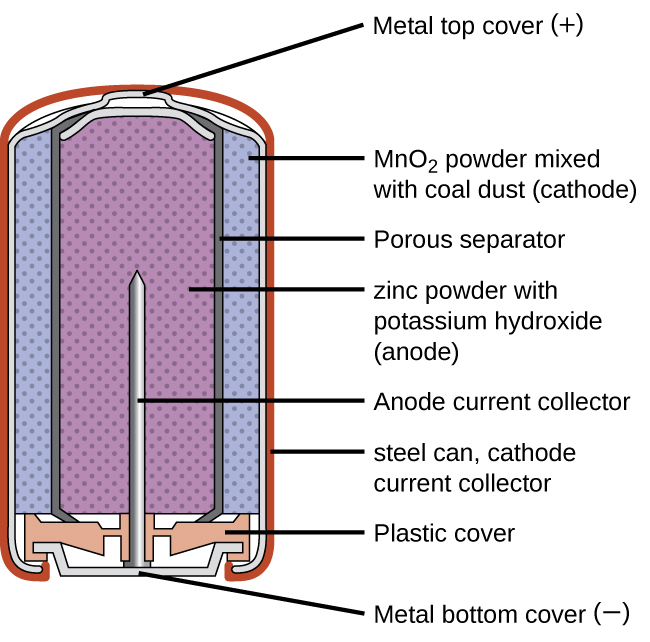
The voltage of a single cell is about 1.5 volts. The battery can be reused ad recharged many times. A secondary battery is a battery that can be recharged by regenerating the electrodes inside the cell. When the electrodes are fully consumed after much use, the battery can no longer generate electricity. When the battery is connected to a circuit, a chemical reaction takes place within the cell and current flows through the circuit. The electrodes are submersed in the electrolyte. All chemical cells contain three main parts: a positively charged electrode called the cathode a negatively charged electrode, called the anode and a chemical substance, called an electrolyte. Before that time, only static electricity an innovation with no practical value could be produced.Ĭhemical batteries convert chemical energy into electrical energy by means of a chemical reaction, which consumes the metal within the cell. 
The Italian scientist Alessandro Volta invented the first electric battery in about the year 1800. Batteries are used to provide energy for devices from space satellites to trucks, to radios. It is important to remember that the voltage delivered. Batteries are a convenient portable source of energy and they differ from other energy-conversion devices in that batteries contain no moving parts. with an overall cell potential which is initially about 1.5 V, but decreases as the battery is used. Energizer.Ī battery is a device that generates electrical energy. Buy 1.5 V Single Use Batteries and get the best deals at the lowest prices on eBay Great Savings & Free Delivery. Principal Dry Battery Systems and Typical Characteristics. Common lithium primary cells, with a terminal voltage around 3 volts, are not made in sizes interchangeable with 1.5 volt cells. "A new 'D' Battery has an emf of 1.5 V… a current of 28 A is produced"Įnergy Density.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
